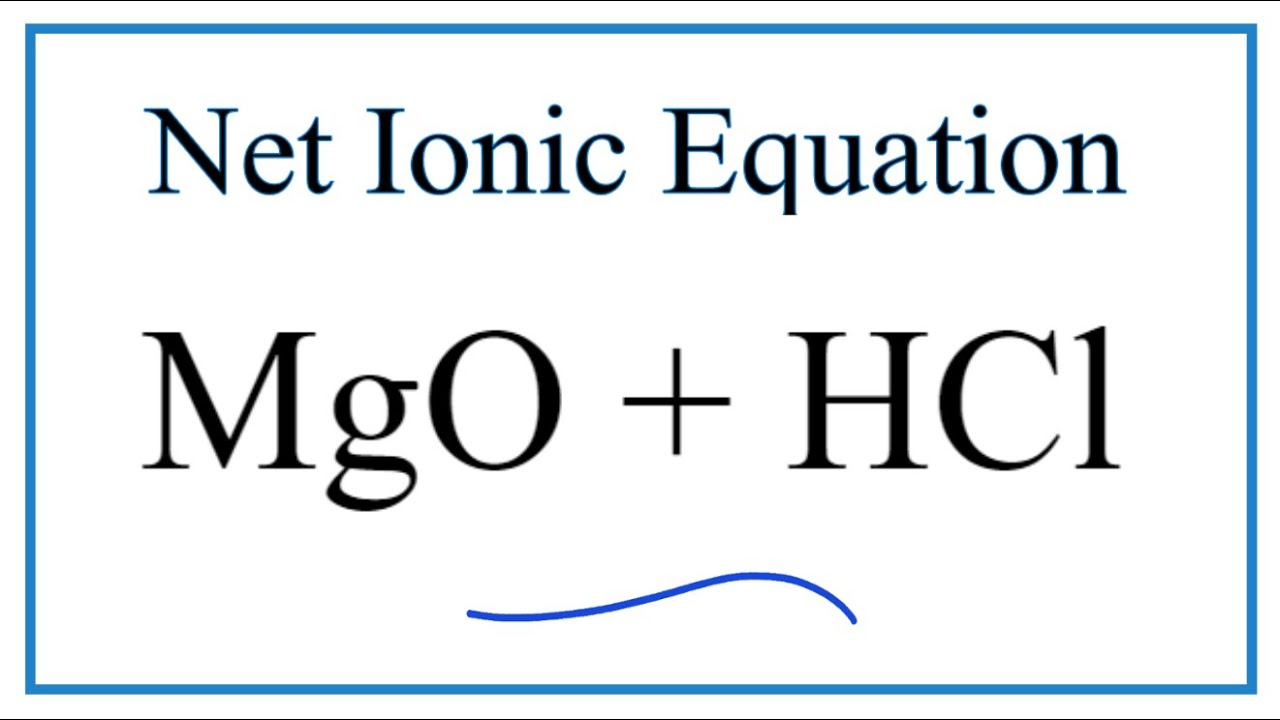16/10/2015 Acids and Bases Hydrochloric acidCitric acidWater An acid is a “proton donor”: A base is a “proton acceptor”: H Cl H +- H O Na H O - + Sodium. - ppt download

Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.

SOLVED: Consider the reaction of magnesium oxide with hydrochloric acid: Mgo (s) 2 HCI (aq) + MgClz (aq) Hz (g) AH -151kI Is the reaction of MgO with HCl exothermic or endothermic?

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download