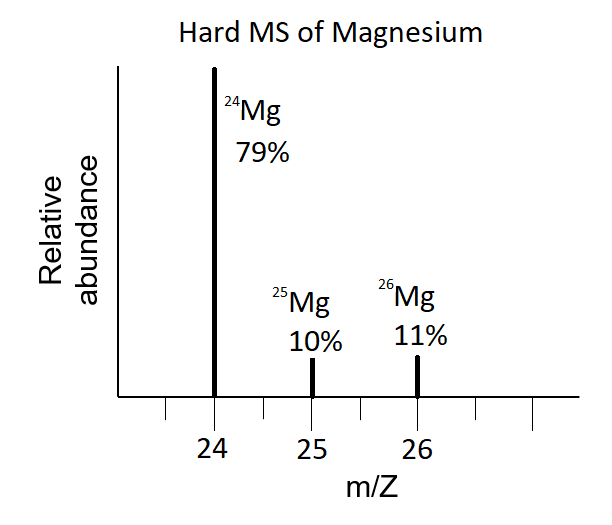
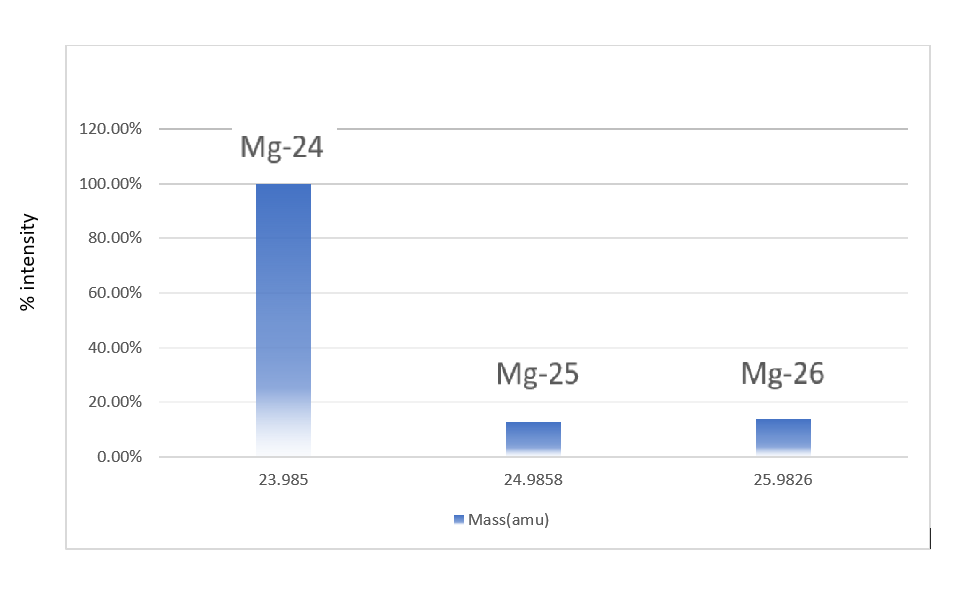
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their

Surface atomic magnesium abundance of magnesium strips after soaking in... | Download Scientific Diagram

Lick index diagram to diagnose the magnesium-to-iron abundance ratio in... | Download Scientific Diagram
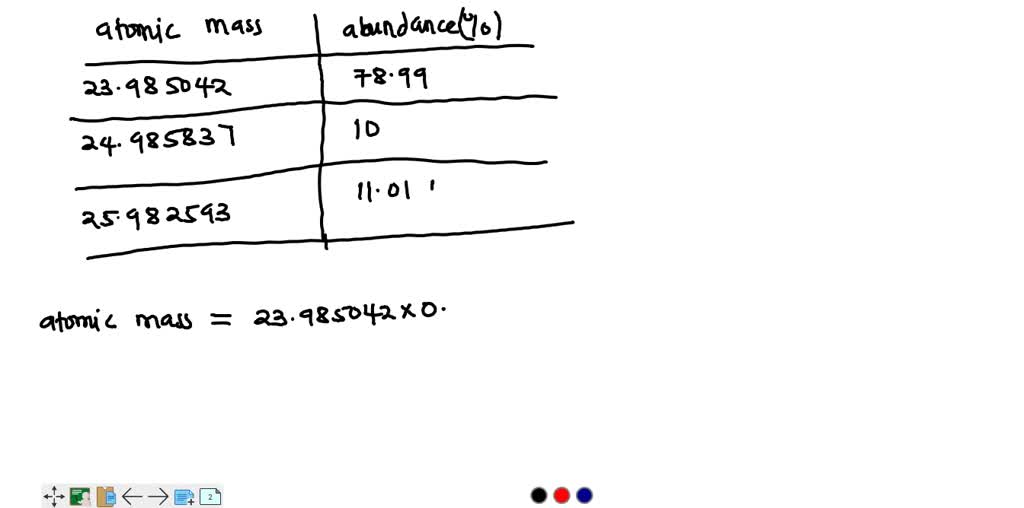
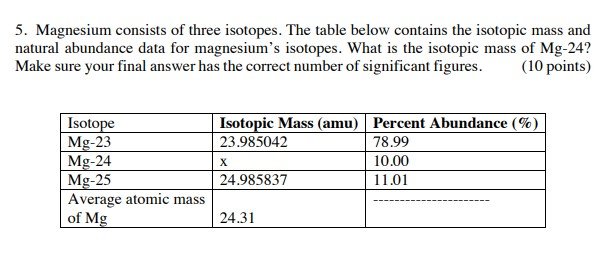
SOLVED: Magnesium has three natural isotopes. Their abundance percentages and masses in nature is as follows: 78.99%; 23.985042 akb, 10.00%; 24.985837 akb, 11.01%; 25.982593 akb. Accordingly, the weighted average atomic mass of magnesium calculate.
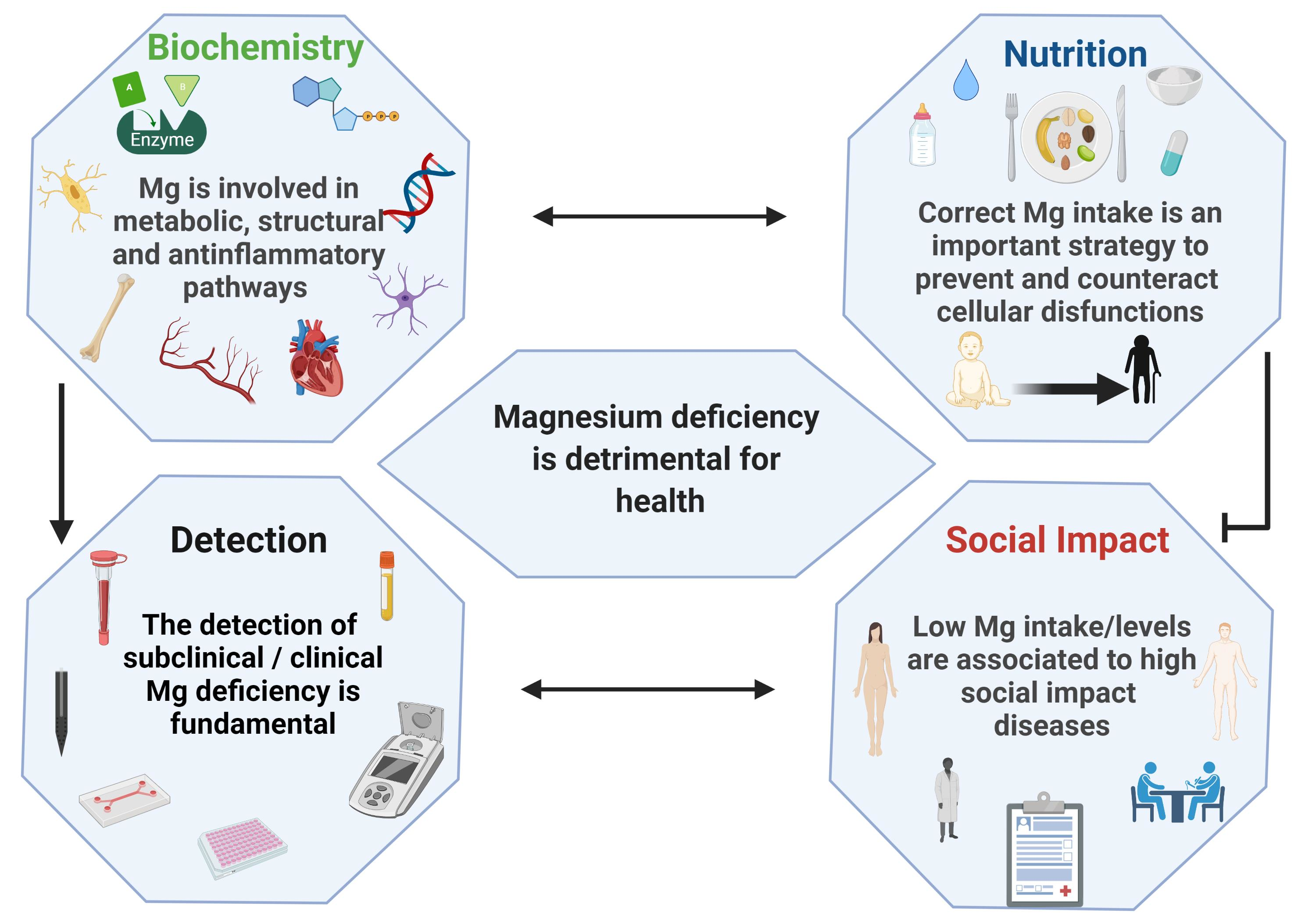
Nutrients | Free Full-Text | Magnesium: Biochemistry, Nutrition, Detection, and Social Impact of Diseases Linked to Its Deficiency

1 Warm Up Isotopes Mass of Isotope Abundance 24 Mg =24.0 amu 78.70% 25 Mg = 25.0 amu 10.13% 26 Mg = 26.0 amu 11.17% Calculate the mass average of magnesium. - ppt download

⚗️Magnesium occurs naturally in only three isotopes. ^24 Mg has an isotopic mass of 23.9850 amu and - Brainly.com